Get Healthy!
Results for search "Food &, Drug Administration".
Health News Results - 435
The U.S. Food and Drug Administration’s recent decision to withhold approval of a new skin cancer treatment fell like a hammer on doctors who treat melanoma and patients who saw that the drug had prolonged the lives of a third of the participants in a clinical trial.
“It was devastating news,” sai...
- Arthur Allen and KFF Health News HealthDay Reporters
- |
- May 16, 2026
- |
- Full Page
After months of pressure from the Trump administration, U.S. Food and Drug Administration (FDA) Commissioner Dr. Marty Makary has quit.
President Donald Trump posted the news on
The U.S. Food and Drug Administration (FDA) has approved Bizengri to treat an ultra-rare, aggressive cancer that forms in the bile ducts.
Bizengri (zenocutuzumab-zbco) is the first drug approved for adults with advanced, inoperable or metastatic cholangiocarcinoma with NRG1 gene fusion, the FDA announced.
NRG1, or neuregulin 1, gene...
- Andria Park Huynh HealthDay Reporter
- |
- May 11, 2026
- |
- Full Page
For the first time ever, the U.S. Food and Drug Administration (FDA) authorized the sale of fruit-flavored e-cigarettes ("vapes") for adults 21 and over.
The move comes amid news of President Donald Trump reportedly pressing FDA Commissioner Dr. Ma...
- Andria Park Huynh HealthDay Reporter
- |
- May 6, 2026
- |
- Full Page
The U.S. Food and Drug Administration (FDA) granted expanded access for the use of an experimental pancreatic cancer drug, daraxonrasib.
This means the drug will be available for early access to those who previously received conventional treatment for metastatic pancreatic ductal adenocarcinoma (PDAC). A healthcare provider must request access to the medication for their patient, accordi...
- Andria Park Huynh HealthDay Reporter
- |
- May 4, 2026
- |
- Full Page
A new proposal could make it easier for patients to access breakthrough medical devices through Medicare.
On Thursday, federal regulators announced a plan they’re calling RAPID.
The goal? To better coordinate how the U.S. Food and Drug Administration (FDA) and Medicare review these new innovative devices. FDA determines whether devices are safe, and Medicare decides whether t...
- HealthDay Staff HealthDay Reporter
- |
- April 24, 2026
- |
- Full Page
The U.S. Food and Drug Administration (FDA) will soon review whether certain peptides should be allowed in customized medications made by compounding pharmacies.
Peptides are small chains of amino acids that are marketed for a wide range of uses, including treating wounds, obesity,
New anti-amyloid drugs approved to treat Alzheimer’s disease have no clinically meaningful positive effects for patients, a major evidence review has concluded.
Drugs like Leqembi (lecanemab) and...
- Dennis Thompson HealthDay Reporter
- |
- April 16, 2026
- |
- Full Page
The U.S. Food and Drug Administration (FDA) has told Eli Lilly to study possible heart, liver and other risks tied to its new obesity drug Foundayo, according to an approval letter released Tuesday.
- HealthDay Staff HealthDay Reporter
- |
- April 15, 2026
- |
- Full Page
A new daily pill to help with weight loss has been approved by the U.S. Food and Drug Administration (FDA), and it moved through review faster than most drugs in recent years.
The drug, called Founda...
- HealthDay Staff HealthDay Reporter
- |
- April 2, 2026
- |
- Full Page
Two chocolate products marketed for sexual enhancement are being recalled because they were found to contain hidden prescription drug ingredients.
The recall involves "Gold Lion Aphrodisiac Chocolate" and "ilum Sex Chocolate," sold by the company Gear Isle, according to the U.S. Food and Drug Administration (FDA).
Officials said the products c...
- HealthDay Staff HealthDay Reporter
- |
- March 31, 2026
- |
- Full Page
The U.S. Food and Drug Administration (FDA) has warned a biotech company about claims that its bladder cancer drug could treat and prevent multiple types of cancer.
The agency sent a warning letter Tuesday to ImmunityBio, saying recent statements about its drug Anktiva were misleading.
The concer...
- HealthDay Staff HealthDay Reporter
- |
- March 26, 2026
- |
- Full Page
Health officials are warning consumers about a cream cheese recall that has been raised to the most critical level due to possible bacterial contamination.
The U.S. Food and Drug Administration (FDA) said certain cream c...
- HealthDay Staff HealthDay Reporter
- |
- March 17, 2026
- |
- Full Page
The U.S. Food and Drug Administration (FDA) may allow some flavored e-cigarettes back on the market, but there’s a catch.
They would be marketed to adults, not teens.
Under guidance released Monday, the FDA said it may consider approving vape flavors such as mint, coffee, tea and spices like clove or cinnamon. But it will continue rejecting sweet or fruit-flavored products, wh...
- HealthDay Staff HealthDay Reporter
- |
- March 11, 2026
- |
- Full Page
The U.S. Food and Drug Administration (FDA) has approved a generic drug for a very rare brain disorder, but said it should not be considered a treatment for autism.
On Tuesday, the agency cleared leucovorin for people with a genetic condition t...
- HealthDay Staff HealthDay Reporter
- |
- March 11, 2026
- |
- Full Page
Dr. Vinay Prasad, who leads the U.S. Food and Drug Administration (FDA) division that oversees vaccines and complex medical treatments, is leaving the agency at the end of April.
Prasad took on the job last May but faced criticism during his short stint.
FDA Commissioner
The U.S. Food and Drug Administration’s (FDA) chief says the agency will begin offering bonuses to drug reviewers who complete their work ahead of schedule.
Dr. Marty Makary described the effort as a pilot program during a staff meeting last week. The first quarterly bonus payments could begin going out in Au...
- HealthDay Staff HealthDay Reporter
- |
- March 2, 2026
- |
- Full Page
U.S. health officials are proposing a new way to develop and approve custom-made treatments for people with rare and hard-to-treat conditions.
The U.S. Food and Drug Administration (FDA) just released a draft of guidelines that would create a special pathway for therapies designed for just a small number of people. Drug companies often avoid these, because they are considered unprofitable...
- I. Edwards HealthDay Reporter
- |
- February 24, 2026
- |
- Full Page
In a major blow to vaccine development, the U.S. Food and Drug Administration (FDA) said it will not review Moderna’s application for the first mRNA-based flu shot.
Dr. Vinay Prasad, the nation’s top vaccine regulator, told the company it lacked an "adequate and well-controlled" study, Moderna<...
- Carole Tanzer Miller HealthDay Reporter
- |
- February 12, 2026
- |
- Full Page
The U.S. Food and Drug Administration (FDA) is taking a fresh look at the safety of a chemical preservative found in many packaged foods.
The agency announced it has launched a full review to decide whether butylated hydroxyanisole, or BHA, is still safe to use in food and food packaging based on the latest science.
As part of that process, the FDA is asking the public to submit new...
- I. Edwards HealthDay Reporter
- |
- February 11, 2026
- |
- Full Page
Olympia Provisions has recalled about 1,930 pounds of ready-to-eat holiday sausage.
The recalled meat is wrapped and vacuum-sealed in 16-ounce clear pouches and labeled “OLYMPIA PROVISIONS UNCURED HOLIDAY KIELBASA.”
The U.S. Department of Agriculture’s Food Safety and Inspection Service (FSIS)
The U.S. Food and Drug Administration (FDA) is weighing a change that could make warning labels on dietary supplements appear less often on packaging.
Unlike prescription drugs, dietary supplements are not reviewed by the FDA for safety or effectiveness before they are sold.
Instead, a 1994 federal law requires companies to include a disclaimer whenever they make health claims, such...
- I. Edwards HealthDay Reporter
- |
- December 19, 2025
- |
- Full Page
THURSDAY, Dec. 18, 2025 (HealthDay News -- The U.S. Food and Drug Administration (FDA) approved a new safety warning for Depo-Provera, a widely used birth control shot made by Pfizer, alerting patients to a possible risk for a type of brain tumor called meningioma.
The agency signed off last week o...
- I. Edwards HealthDay Reporter
- |
- December 18, 2025
- |
- Full Page
Americans may soon have access to a new sunscreen ingredient already used around the world.
The U.S. Food and Drug Administration (FDA) announced Dec. 11 that it is reviewing a proposal to allow bemotrizinol in sunscreens sold in the United States.
The ingredien...
- I. Edwards HealthDay Reporter
- |
- December 12, 2025
- |
- Full Page
An at-home device that sends a gentle electrical current to the brain to help treat depression has been cleared by the U.S. Food and Drug Administration (FDA).
Experts say the move could expand access to care for many folks.
The prescription headset, made by Sweden-based <...
- I. Edwards HealthDay Reporter
- |
- December 12, 2025
- |
- Full Page
The U.S. Food and Drug Administration (FDA) is facing even more leadership changes as drug regulator Dr. Richard Pazdur prepares to retire at the end of the month, the agency confirmed this week.
Pazdur, who has worked at the FDA for 26 years, told senior leaders on Tuesday that he plans to step down, just weeks a...
- I. Edwards HealthDay Reporter
- |
- December 4, 2025
- |
- Full Page
The U.S. Food and Drug Administration (FDA) is tightening restrictions on a gene therapy used to treat Duchenne muscular dystrophy after two teenagers died from liver failure linked to the medication.
The FDA decision limits the use of Elevidys, made by Sarepta Therapeutics, to boys who are 4 years an...
- I. Edwards HealthDay Reporter
- |
- November 17, 2025
- |
- Full Page
The U.S. Food and Drug Administration (FDA) has appointed one of its most respected cancer drug regulators to lead the agency’s main division for approving new drugs.
The appointment of Dr. Richard Pazdur comes after a turbulent year with hundreds of staff departures within the agency.
Pazdur, who has ...
- I. Edwards HealthDay Reporter
- |
- November 13, 2025
- |
- Full Page
The U.S. Food and Drug Administration (FDA) is eliminating the prominent "black box" warnings on many hormone replacement therapy (HRT) medications, signaling a major shift in how the treatment is viewed for menopausal women.
The decision affects products containing estrogen or progestogen, alone or combined, that are prescribed to treat troublesome menopause symptoms like hot flashes, mo...
- Deanna Neff HealthDay Reporter
- |
- November 12, 2025
- |
- Full Page
The head of the U.S. Food and Drug Administration’s (FDA) drug division has resigned while under internal investigation, saying he was pushed out after raising concerns about how the agency planned to fast-track some new drugs.
Dr. George Tidmarsh, who joined the FDA in July, stepped down Sunday, The New York T...
- I. Edwards HealthDay Reporter
- |
- November 4, 2025
- |
- Full Page
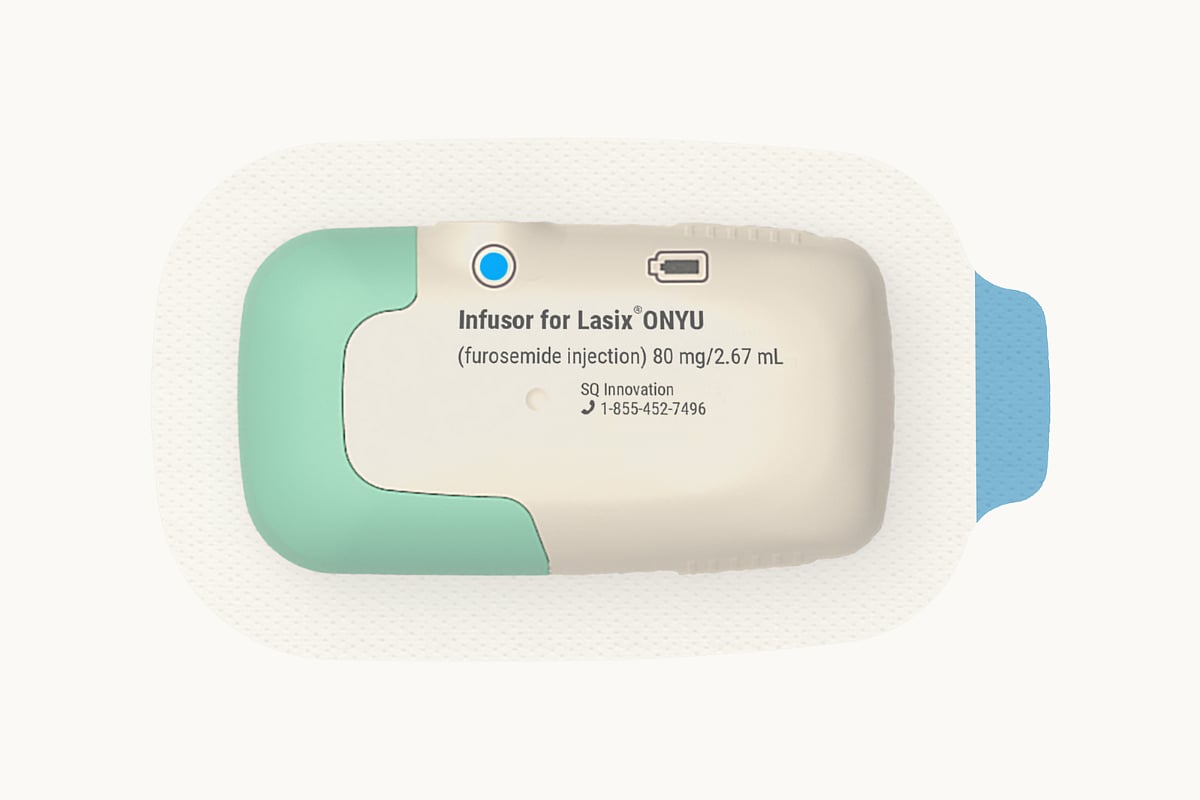
A new at-home version of a common heart failure drug could make treatment easier for millions of Americans.
The U.S. Food and Drug Administration (FDA) has approved Lasix ONYU (furosemide injection), a new drug-device combination developed by SQ Innovation, Inc., for treating edema caused by chronic
For the first time, people can get their annual flu vaccine without leaving the comfort of home.
FluMist, a nasal spray flu vaccine made by AstraZeneca, is now available for at-home use through a service called FluMist Home, the company announced.
The U.S. Food and Drug Administration (FDA) first approved FluMist in 2003 for use in doctors&rsqu...
- I. Edwards HealthDay Reporter
- |
- August 19, 2025
- |
- Full Page
The U.S. Food and Drug Administration (FDA)’s top vaccine regulator is returning to his post less than two weeks after the White House had him ousted.
Dr. Vinay Prasad will again head the FDA’s Center for Biologics...
- I. Edwards HealthDay Reporter
- |
- August 11, 2025
- |
- Full Page
The U.S. Food and Drug Administration’s (FDA) top vaccine official is stepping down after just three months in a role that upset drug companies, patient groups and some political leaders.
Dr. Vinay Prasad will leave the agency to “spend more time with his family,” a spokesperson for the U.S. Department of Health and Human Service...
- HealthDay Reporter
- I. Edwards
- |
- July 31, 2025
- |
- Full Page
Ritz peanut butter cracker sandwiches are being recalled due to the possible presence of undeclared peanuts, a major allergen.
The affected products contain individually wrapped packs that may have been mislabeled as cheese sandwiches instead of peanut butter ones, according to Newsweek. The outer cartons were labeled correctly and indicated the presence of peanuts.
Mondel&...
- HealthDay Reporter
- Denise Mann
- |
- July 25, 2025
- |
- Full Page
The U.S. Food and Drug Administration (FDA) has upgraded a recall of a commonly prescribed thyroid medication due to what it described as "subpotent" active ingredients.
The recall of more than 160,000 bottles of levothyroxine sodium, which went into effect June 20, was upgraded to a Class II recall on July 23. Class II recalls occur ...
- HealthDay Reporter
- Denise Mann
- |
- July 24, 2025
- |
- Full Page
WEDNESDAY, July 23, 2025 (HealthDay News) --Two types of tuna sold in seven states are being recalled due to listeria concerns.
The first recall affects tuna salad and ready-to-eat foods containing tuna salad from Beaverton, Oregon-based Reser's Fine Foods. The products were sold at ...
- HealthDay Reporter
- Denise Mann
- |
- July 23, 2025
- |
- Full Page
More than 67,000 cases of Power Stick deodorant have been recalled due to an undisclosed manufacturing issue.
The recalled deodorants, made by A.P. Deauville of Easton, Pa., did not fully comply with federal product safety standards, according to the U.S. Food and Drug Administration (FDA).
In issuing
Thousands of health workers lost their jobs this week after a U.S. Supreme Court ruling cleared the way for the Trump administration to move forward with major staffing cuts.
On Monday, the U.S. Department of Health and Human Services (HHS) finalized 10,000 layoffs across federal h...
- HealthDay Reporter
- I. Edwards
- |
- July 16, 2025
- |
- Full Page
The U.S. Food and Drug Administration (FDA) says it plans to use artificial intelligence (AI) to help speed the approval of new drugs and medical devices.
That's one of several priorities federal officials detailed June 10 in JAMA.
They said AI could help shorten review times, speeding delivery of treat...
- HealthDay Reporter
- I. Edwards
- |
- June 11, 2025
- |
- Full Page
Salmonella-tainted tomatoes in three southern states could cause severe illness or even death, the U.S. Food and Drug Administration (FDA) warns.
The FDA has updated an ongoing recall of tomatoes distributed in Georgia, North Carolina and South Carolina to Class I -- its highest warning level, The New York Times reported.
A Class I recall means there is a reasonable chance ...
- HealthDay Reporter
- I. Edwards
- |
- June 2, 2025
- |
- Full Page
Publix is recalling one of its popular GreenWise baby food pouches because it may contain lead, the company said this week.
The recalled product is the Pear, Kiwi, Spinach & Pea Baby Food pouch.
The supermarket chain said it found the issue through routine testing a...
- HealthDay Reporter
- I. Edwards
- |
- May 20, 2025
- |
- Full Page
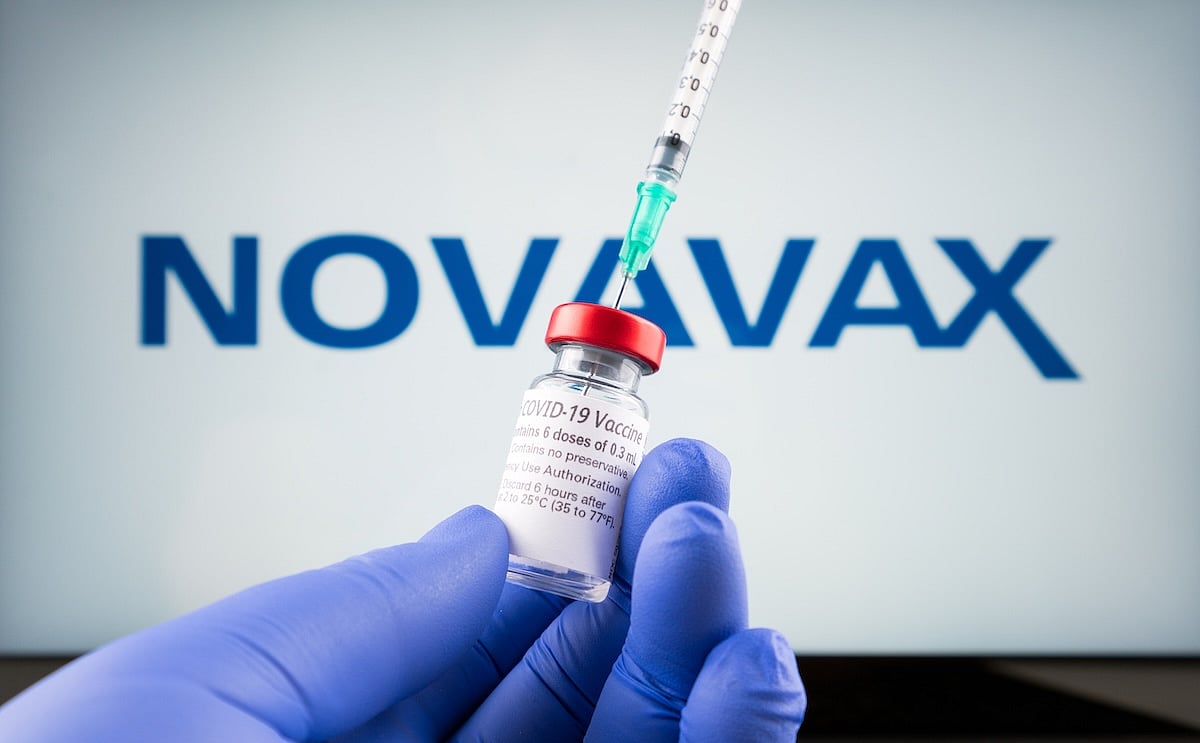
The U.S. Food and Drug Administration (FDA) has granted full approval to Novavax’s COVID-19 vaccine, but only for certain people.
The vaccine is now approved for adults ages 65 and older, or for people ages 12 to 64 who have at least one health condition that puts them at higher risk of serious illness from
Women now have a new way to check their risk for cervical cancer -- from the comfort of their own home.
The U.S. Food and Drug Administration (FDA) has approved the Teal Wand, an at-home test that screens for human papillomavirus (HPV), the virus tha...
- HealthDay Reporter
- I. Edwards
- |
- May 12, 2025
- |
- Full Page
The U.S. Food and Drug Administration (FDA) is warning consumers and tattoo artists that two tattoo inks have tested positive for harmful bacteria and could lead to serious infections.
The affected products are:
Sacred Tattoo Ink, Raven Black (CI# 77266; Lot#: RB0624, Best Before: June 28, 2027)
Sacred Tattoo Ink, Sunny Daze (CI# 21095; Lot#: SD1124, Best Befo...
- HealthDay Reporter
- I. Edwards
- |
- May 9, 2025
- |
- Full Page
The U.S. Food and Drug Administration (FDA) has chosen Dr. Vinay Prasad, a professor at the University of California-San Francisco, to lead its Center for Biologics Evaluation and Research.
The division oversees vaccines and biologic medicines, including gene therapies, CNN reported.
Prasad is a hematologist-oncologist, a specialis...
- HealthDay Reporter
- I. Edwards
- |
- May 7, 2025
- |
- Full Page
The official in charge of federal food and drug safety inspections will retire May 14.
Michael Rogers, associate commissioner for inspections and investigations at the U.S. Food and Drug Administration (FDA), announced his decisio...
- HealthDay Reporter
- I. Edwards
- |
- May 7, 2025
- |
- Full Page