Get Healthy!
Staying informed is also a great way to stay healthy. Keep up-to-date with all the latest health news here.
03 Apr
A Good Cry Isn’t Always Good for You, Study Finds
Tears don’t automatically bring relief—and can sometimes make you feel worse, according to a new study. It all depends on why you’re crying.
02 Apr
Occasional Heavy Drinking May Triple Risk of Liver Damage, Study Finds
New research finds even occasional heavy drinking — just once a month — may raise the risk of serious liver scarring, even in otherwise moderate drinkers.
01 Apr
Study Questions Effectiveness of KT Tape for Pain Relief
A large review of more than 300 studies finds KT tape may offer short-term pain relief, but its overall benefits remain uncertain. Researchers say it shows little impact on strength, mobility, or long-term outcomes.
What Sea Creatures Reveal About How Fast People Age
This is no ordinary fish story.
Researchers who videotaped every moment in the lives of 81 African turquoise killfish gleaned intriguing insights into the aging process that may also apply to humans.
Though killfish live just four to eight months, they share important biological features with people, including a complex brain. That m...
- Carole Tanzer Miller HealthDay Reporter
- |
- April 5, 2026
- |
- Full Page
How to Tell if Spring Symptoms Owe to Allergy, Cold or Something More Serious
When your head is splitting, your nose is running nonstop, your eyes are itching and you’re coughing, coughing, coughing, what’s the best way to fight back?
That depends on what’s causing your symptoms, said Dr. Ian Tullberg, an urgent care and family medicine provider at UCHealth in Colorado Springs, Colorado.
It m...
- Carole Tanzer Miller HealthDay Reporter
- |
- April 4, 2026
- |
- Full Page
USDA Warns of Lead Risk in Frozen Dino-Shaped Chicken Nuggets
Food safety officials are warning the public not to eat certain frozen chicken nuggets after tests found dangerous levels of lead.
The U.S. Department of Agriculture Food Safety and Inspection Service (FSIS) issued a public health alert for dinosaur-shaped, ready-to-eat chicken nuggets sold under the Great Value brand at Walmart.
A r...
- HealthDay Staff HealthDay Reporter
- |
- April 3, 2026
- |
- Full Page
New Heart Diet Advice Counters U.S. Guidance on Meat and Dairy
The nation's leading heart health organization is urging folks to rethink where they get their protein.
The goal? Focus more on plants.
The American Heart Association (AHA) released new nutrition guidance recommending plant-based proteins like beans, lentils and nuts instead of red and processed meats.
The group also reco...
- HealthDay Staff HealthDay Reporter
- |
- April 3, 2026
- |
- Full Page
Peeled Garlic Recalled Over Risk of Deadly Botulism
Some peeled garlic is being recalled because it may pose a risk for a serious type of food poisoning.
Tops Friendly Markets announced the recall of Christopher Ranch and Garland brand peeled garlic because the products may be contaminated with Clostridium botulinum, a type of bacteria that can cause life-threatening illness.
- HealthDay Staff HealthDay Reporter
- |
- April 3, 2026
- |
- Full Page
Some CDC Lab Testing Paused Amid Internal Review
The U.S. Centers for Disease Control and Prevention (CDC) has paused more than two dozen types of lab tests.
Officials said the move is temporary.
The explanation?
"A routine review to uphold our commitment to high quality laboratory testing," Andrew Nixon, a spokesperson at the U.S. Department of Health and Human Services, sai...
- HealthDay Staff HealthDay Reporter
- |
- April 3, 2026
- |
- Full Page
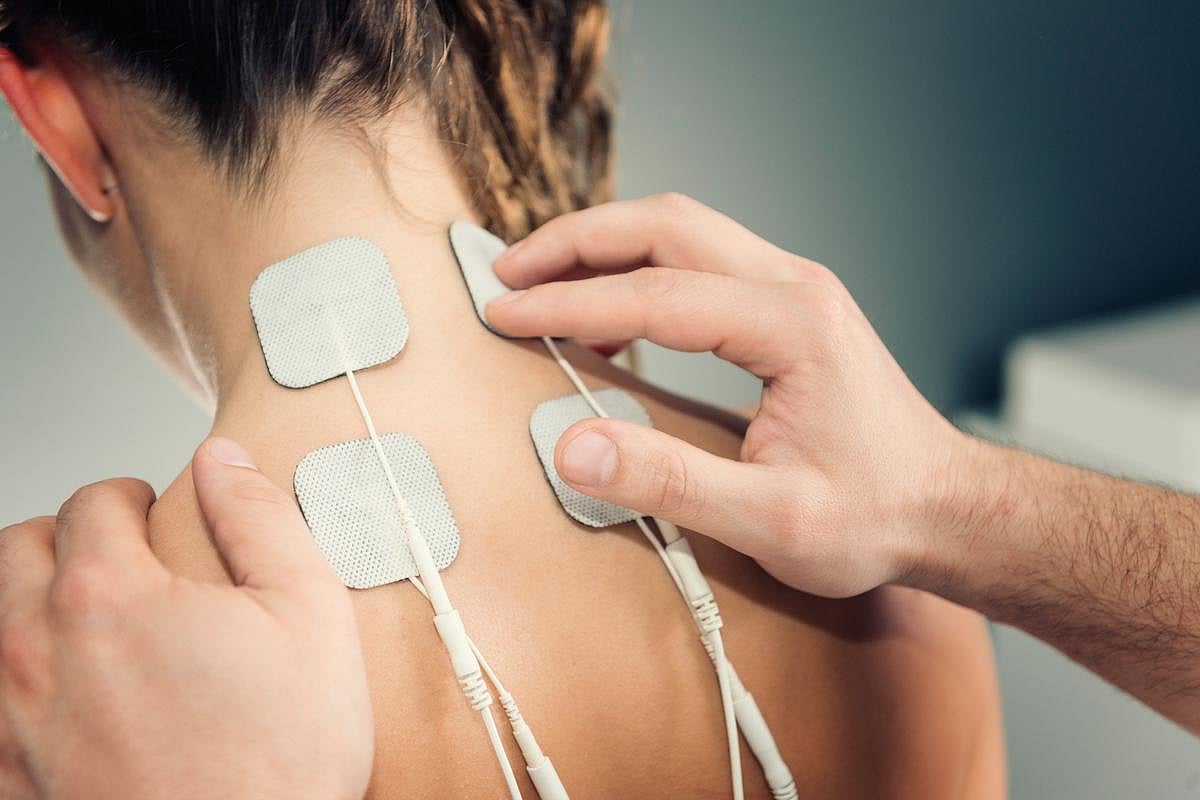
Nerve Stimulation Therapy May Ease Fibromyalgia Pain, Fatigue
In its first "real-world" trial, the nerve stimulation treatment called TENS appeared to reduce the pain and tiredness of fibromyalgia.
"The study shows that TENS provides an added benefit on top of any relief from other treatments," said study first author Dana Dailey of the University of Iowa.
"All the study participants were...
- Ernie Mundell HealthDay Reporter
- |
- April 3, 2026
- |
- Full Page
Psychotherapists Often Poorly Trained in Treating Muscle-Linked Disorders in Males
Boys and men can also develop eating disorders, and rates for these psychiatric issues are rising — even as psychotherapists admit they have trouble spotting and treating them.
So finds a new study of 259 American and Canadian psychotherapists.
"Boys and men with eating disorders are often hiding in plain sight," said study lea...
- Ernie Mundell HealthDay Reporter
- |
- April 3, 2026
- |
- Full Page
Missing From Most Doctor-Patient Talks: Sleep Issues
Even though most folks know it's key to good health, sleep often goes undiscussed during doctor visits, a new survey finds.
Almost half (45%) of adults said they never talk about sleep quality with their doctor, according to a poll from the American Academy of Sleep Medicine (AASM).
That could bring real harm, said AASM spokesp...
- Ernie Mundell HealthDay Reporter
- |
- April 3, 2026
- |
- Full Page
Plastics Chemical Linked To Nearly 2 Million Preterm Births Each Year
A common chemical that makes plastics more pliable may come with a grim downside: Nearly 2 million premature births per year, new research shows.
Di-2-ethylhexylphthalate (DEHP) belongs to a class of chemicals called phthalates, which have long been linked to health hazards in people.
Phthalates are ubiquitous in products rangi...
- Ernie Mundell HealthDay Reporter
- |
- April 3, 2026
- |
- Full Page
Most Americans Don't Realize Brain Donation Is Needed to Study Autism
Americans are overwhelmingly supportive of autism research, but a new survey has uncovered a lack of awareness that could be slowing scientific progress.
Very few realize that the most critical tool for researchers — the human brain — is in short supply.
While nearly everyone agrees that studying the brain is ...
- Deanna Neff HealthDay Reporter
- |
- April 3, 2026
- |
- Full Page
Weekend Binge Drinking Triples Risk of Permanent Liver Damage
Many folks think it’s OK to belt back a few extra rounds on Saturday night if they stay mostly sober during the week.
But saving up your drinks for a single sitting could be a recipe for liver problems, new research suggests.
A team at the University of Southern California’s Keck School of Medicine linked occasional heavy...
- Deanna Neff HealthDay Reporter
- |
- April 3, 2026
- |
- Full Page
FDA Recalls Wawa Milk Over Possible Plastic Contamination
Milk sold at convenience stores in four states is being recalled amid concerns about possible plastic contamination.
The U.S. Food and Drug Administration (FDA) said the recall affects 16-ounce plastic bottles of Wawa milk sold in Delaware, Maryland, New Jersey and Pennsylvania.
The issue was discovered after "foreign plastic materia...
- HealthDay Staff HealthDay Reporter
- |
- April 2, 2026
- |
- Full Page
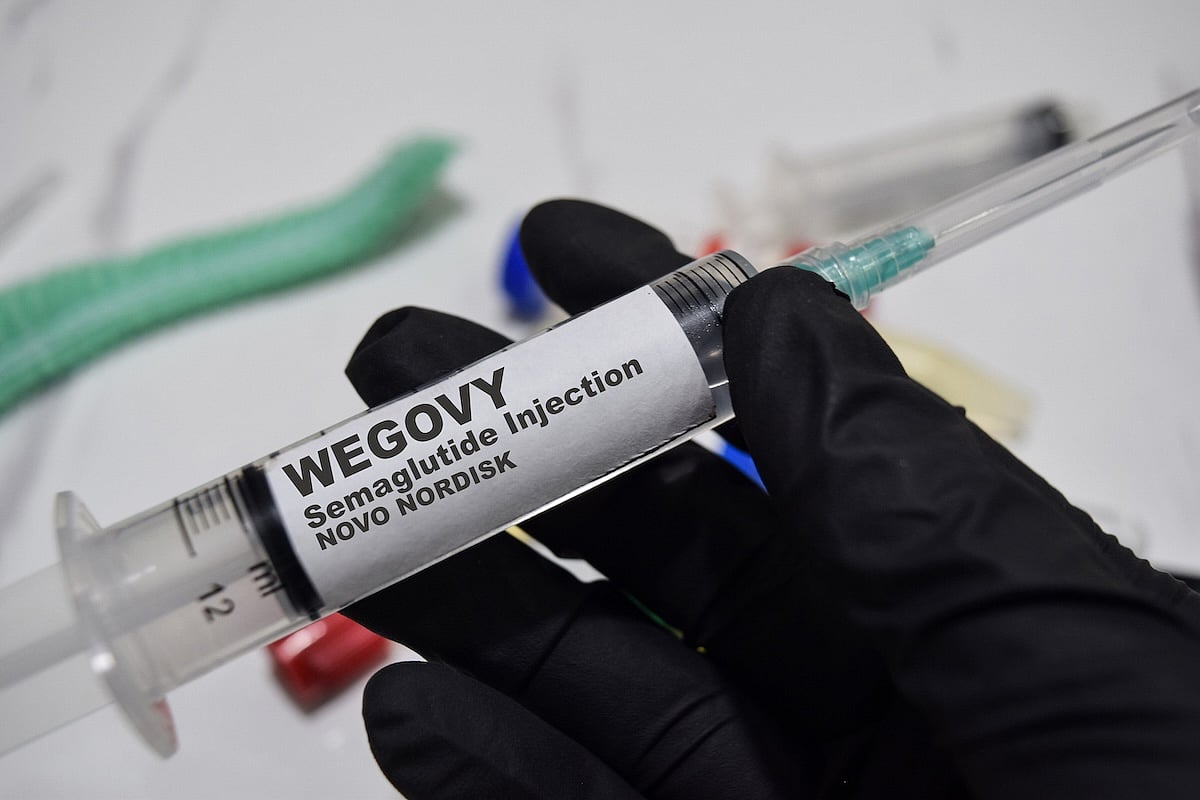
Wegovy Maker Launches Lower-Cost Subscription Plans
A popular weight loss drug may soon be more affordable for some people.
Danish pharmaceutical company Novo Nordisk has launched a subscription program for its obesity treatment Wegovy, offering lower and more predictable monthly prices.
The program is aimed at those who pay for the medication themselves, The Wall Street Journal
- HealthDay Staff HealthDay Reporter
- |
- April 2, 2026
- |
- Full Page
FDA Approves New Weight Loss Pill in Record Time
A new daily pill to help with weight loss has been approved by the U.S. Food and Drug Administration (FDA), and it moved through review faster than most drugs in recent years.
The drug, called Foundayo and developed for Eli Lilly, was approved in just 50 days. That’s 294 days ahead of schedule.
It is the fastest approval of a n...
- HealthDay Staff HealthDay Reporter
- |
- April 2, 2026
- |
- Full Page
Lawsuit Over Viral David Protein Bars Dropped Without Explanation
A lawsuit claiming popular protein bars had misleading nutrition labels has been dropped, but questions about the bars remain.
The case targeted David bars, which have gained a massive amount of attention online, especially among health and wellness influencers.
Filed in January, the lawsuit claimed independent lab tests showed the b...
- HealthDay Staff HealthDay Reporter
- |
- April 2, 2026
- |
- Full Page
Pandemic Spurred Increase In Screen Time Among Children, Teens
The COVID-19 pandemic fueled a dramatic rise in screen use among children and teenagers, a new evidence review says.
Kids spent more time with screens after the pandemic, using computers, video games, smartphones and tablets at rates higher than before COVID struck, researchers recently reported in the journal Clinical Child Psychology...
- Dennis Thompson HealthDay Reporter
- |
- April 2, 2026
- |
- Full Page
Siblings Crucial To Middle-Aged People Grieving The Loss Of A Parent, Study Says
Having more brothers and sisters might make it easier for middle-aged folks to cope with the death of a parent, especially a mother, a new study says.
Middle-aged adults needed fewer antidepressants and other mental health drugs before and after a parent’s death if they were supported by a network of siblings, researchers reported Ma...
- Dennis Thompson HealthDay Reporter
- |
- April 2, 2026
- |
- Full Page
New Rapid Urine Test Could Revolutionize Treatment of UTIs
A new rapid urine test could lead to more targeted and effective treatment of urinary tract infections (UTI), researchers say.
It currently takes labs two to three days to determine which antibiotic would work best against an individual’s UTI.
But the new test can turn around results in just under six hours, creating the potent...
- Dennis Thompson HealthDay Reporter
- |
- April 2, 2026
- |
- Full Page
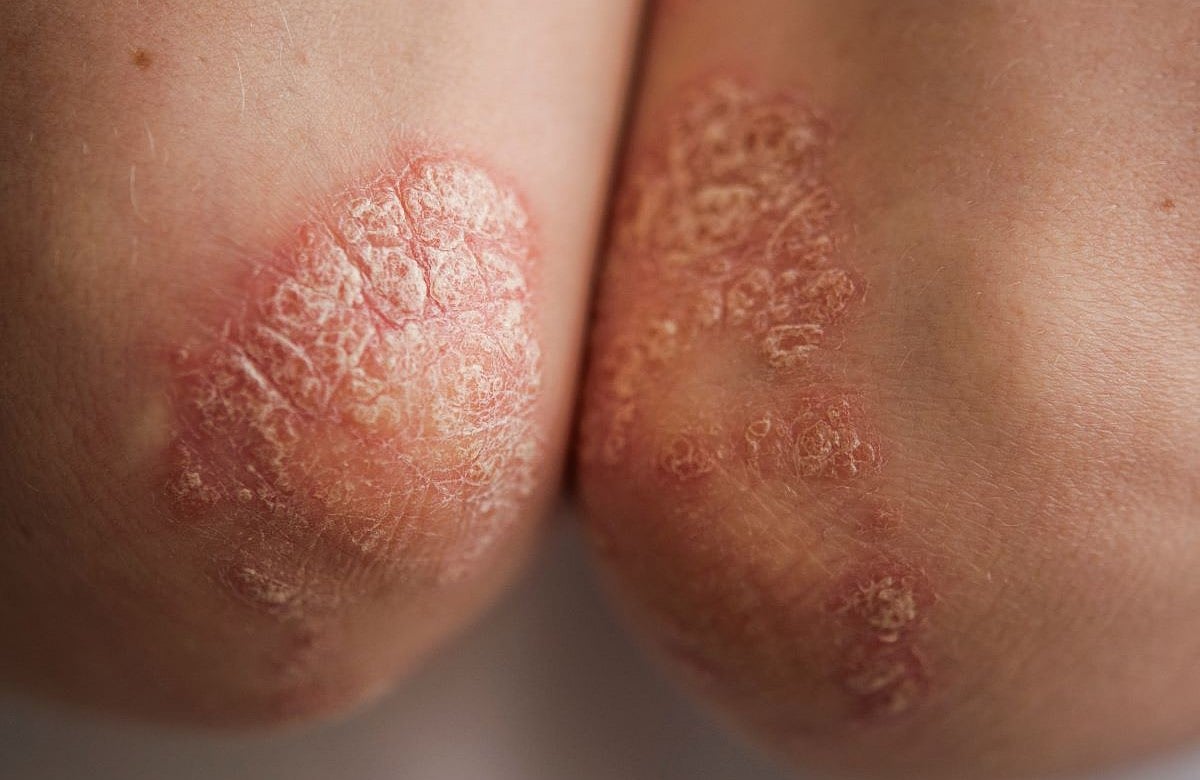
New Pill Could Change Plaque Psoriasis Treatment
Folks with severe plaque psoriasis often have to choose between convenient pills that don’t work very well or highly effective injections that come with the hassle of needles.
That trade-off may soon change. New clinical trial data suggests a once-daily pill called zasocitinib may provide the clear skin once only expected from shots....
- HealthDay Staff HealthDay Reporter
- |
- April 2, 2026
- |
- Full Page